Lithium 7
Click any image to download full size.


The left model has the neutron shell on top, the right model uses the same shells, but
stacks the proton shell on top. Note the single hybridized P.V.B. on the right model.
Lithium 7 (P3-20P) With the Neutron (N4-22P) Shell On Top








 This stacked lithium model was produced from stacking shell P3-20P
and N4-22P,
with the P3 shell nestled inside the N4 shell. As you can see, with the proton
shell taking the inner position, two normal orbital lobes are generated by the
PVB's. Inverting the stacking order would generate a single hybridized
orbital via linearization of one of the PVB's, assuming the shells maintain
their polar relationship with each other as shown. I'm rather sorry I did not
build the model with the proton shell on top, as I'm beginning to think the
proton shell most frequently takes the outer position.
This stacked lithium model was produced from stacking shell P3-20P
and N4-22P,
with the P3 shell nestled inside the N4 shell. As you can see, with the proton
shell taking the inner position, two normal orbital lobes are generated by the
PVB's. Inverting the stacking order would generate a single hybridized
orbital via linearization of one of the PVB's, assuming the shells maintain
their polar relationship with each other as shown. I'm rather sorry I did not
build the model with the proton shell on top, as I'm beginning to think the
proton shell most frequently takes the outer position.
 This model relies on
a single triangle of complementary quarks to lock the shells in place, shown
in figures 1 and 2. The opposite pole of the particle (figures 3 & 4) show the
polar triangles of the two shells are rotated 180 degrees with respect to each other.
Figure 7 shows a side view of the atom with a down quark from the P3 shell peeking
out between a down and an up quark in the N4 shell. I think this is going to be a
common feature of isotopes having more neutrons than protons, because it is obvious
that most isotopes have differing numbers of protons and neutrons, and therefore can
not have convenient and aesthetically pleasing complementary shell structures like
the models for Boron 10 and Helium 4.
This model relies on
a single triangle of complementary quarks to lock the shells in place, shown
in figures 1 and 2. The opposite pole of the particle (figures 3 & 4) show the
polar triangles of the two shells are rotated 180 degrees with respect to each other.
Figure 7 shows a side view of the atom with a down quark from the P3 shell peeking
out between a down and an up quark in the N4 shell. I think this is going to be a
common feature of isotopes having more neutrons than protons, because it is obvious
that most isotopes have differing numbers of protons and neutrons, and therefore can
not have convenient and aesthetically pleasing complementary shell structures like
the models for Boron 10 and Helium 4.
 The radical string pinned to the N4 polar triangle in figures 1 & 2 is likely a fixed feature
of the model, but it might be possible to pin the second radical string to a different Up
quark on the N4 shell, yielding a different bond angle.
The radical string pinned to the N4 polar triangle in figures 1 & 2 is likely a fixed feature
of the model, but it might be possible to pin the second radical string to a different Up
quark on the N4 shell, yielding a different bond angle.
Stereograms: Neutron Shell On Top


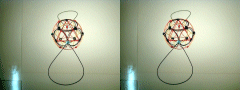


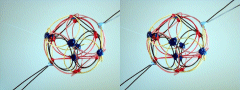
 I went ahead and built another stacked lithium 7 model using the same shells
(P3-20P & N4-22P), but this time stacked them with the proton shell on top. I was in
the process of taking stereograms of the models when I built this, so all I have are
stereograms to show you. As you can see, the "locked" polar triangles of the two shells
spawn a hybridized P.V.B. (the circular pinned string). I'm pretty sure this is the normal
stacking order for these two shells. (See Stereograms below.)
I went ahead and built another stacked lithium 7 model using the same shells
(P3-20P & N4-22P), but this time stacked them with the proton shell on top. I was in
the process of taking stereograms of the models when I built this, so all I have are
stereograms to show you. As you can see, the "locked" polar triangles of the two shells
spawn a hybridized P.V.B. (the circular pinned string). I'm pretty sure this is the normal
stacking order for these two shells. (See Stereograms below.)
Stereograms: Proton Shell On Top







 (05APR02) If I continue with the hypothesis that the "s" orbital is internal to the nuclear shells,
then one electron is available for bonding. If this electron is found on the "p" lobe,
the nucleus will be inclined to participate in a covalent bond. If it is found on the
hybridized P.V.B., then the nucleus will be inclined to participate in metallic "daisy-chain"
bonds. (See Linearization.)
(05APR02) If I continue with the hypothesis that the "s" orbital is internal to the nuclear shells,
then one electron is available for bonding. If this electron is found on the "p" lobe,
the nucleus will be inclined to participate in a covalent bond. If it is found on the
hybridized P.V.B., then the nucleus will be inclined to participate in metallic "daisy-chain"
bonds. (See Linearization.)
Copyright 1999 by Arnold J. Barzydlo
Home
Chemistry Notes
Key to Nuclear Models
Nuclear Shells
Empirical Laws
How to Build Them
Mensuration
Glossary








![]() This model relies on
a single triangle of complementary quarks to lock the shells in place, shown
in figures 1 and 2. The opposite pole of the particle (figures 3 & 4) show the
polar triangles of the two shells are rotated 180 degrees with respect to each other.
Figure 7 shows a side view of the atom with a down quark from the P3 shell peeking
out between a down and an up quark in the N4 shell. I think this is going to be a
common feature of isotopes having more neutrons than protons, because it is obvious
that most isotopes have differing numbers of protons and neutrons, and therefore can
not have convenient and aesthetically pleasing complementary shell structures like
the models for Boron 10 and Helium 4.
This model relies on
a single triangle of complementary quarks to lock the shells in place, shown
in figures 1 and 2. The opposite pole of the particle (figures 3 & 4) show the
polar triangles of the two shells are rotated 180 degrees with respect to each other.
Figure 7 shows a side view of the atom with a down quark from the P3 shell peeking
out between a down and an up quark in the N4 shell. I think this is going to be a
common feature of isotopes having more neutrons than protons, because it is obvious
that most isotopes have differing numbers of protons and neutrons, and therefore can
not have convenient and aesthetically pleasing complementary shell structures like
the models for Boron 10 and Helium 4.![]() The radical string pinned to the N4 polar triangle in figures 1 & 2 is likely a fixed feature
of the model, but it might be possible to pin the second radical string to a different Up
quark on the N4 shell, yielding a different bond angle.
The radical string pinned to the N4 polar triangle in figures 1 & 2 is likely a fixed feature
of the model, but it might be possible to pin the second radical string to a different Up
quark on the N4 shell, yielding a different bond angle.


